 Excited energy states have higher energies and are represented by the notation of n = 2, 3, 4, etc.įor electrons to move to a higher energy state, they must absorb an amount of energy that is equal to the difference between its ground state and the higher energy state. The lowest energy state is called the ground state and is represented by the notation of n = 1.  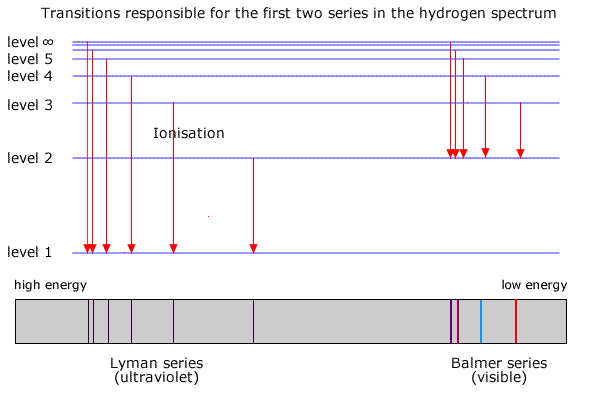
Finally, the absorbed energy results in electron excitement, which causes electrons to move to a higher energy level from the most stable or ground state.Īccording to the Bohr model of hydrogen, electrons in an atom exist in discrete states, which correspond to individual shells or orbitals around the nucleus. Third, the energy causes rotation, which induces the rotation of atoms around the bonds in the molecule. Second, the energy is used in vibration, which causes the distance between the species to change rapidly. First, the energy is used in translation, causing the molecules to move faster. When an atomic or molecular species absorbs energy, the energy is used in four different ways. The emission of colored light from a burning sample is the direct result of the metallic component absorbing energy due to the excitation from the flame and emitting light as a result. This phenomenon is used in fireworks displays, where the color of the fireworks corresponds to a specific characteristic of a metal. Each element has a characteristic light emission when placed into a flame, meaning each element produces a unique color. In this lab, you'll use the metal flame emission test to identify various metals and then analyze the emitted and absorbed light using a spectrophotometer.įlame testing is an analytical technique where a sample is placed in a flame, and the characteristic flame color is used to identify the substance. And since atoms and molecules absorb and release energy differently, the emitted light actually contains a range of wavelengths and intensities forming an emission spectrum.Ī spectrophotometer is used to measure the range of wavelengths of emitted light. Now, a sample actually consists of the metal, metal ions, metal oxides, metal hydroxides, and metal salts. For example, barium will emit a yellowish-green color in the flame, while copper emits blue-green and potassium a pinkish-purple. In the metal flame emission test, we apply a metal sample to a hot flame and observe the color of the emitted light. This emitted wavelength, or color of light, is specific to the atom present and is used to identify a metal sample in the metal flame emission test. The wavelength of the emitted light depends on the energy level that the electron was excited to and the level that it relaxes back to. When the electrons relax back down to a lower state, or the ground state, the excess energy gained is released in the form of emitted light. When an atom absorbs energy, it gets excited, and electrons in the atom move from the ground state to a higher energy level. When an electron is at its lowest possible energy level, it is said to be at its ground state. Atoms have multiple energy levels for their electrons, which are different for each element. But before we dive into the technique further, let's take a step back. Thus, we can use the color of the emitted light to determine the metal present. You can clearly see this phenomenon when watching a fireworks display.ĭifferent metals emit different colors. When sufficient energy is applied to some metals through a hot flame, they emit light with a characteristic wavelength or color. When this technique is used to identify metal species, it is called metal flame emission testing. Flame testing is an analytical technique where a sample is applied to a flame, and the characteristic emission spectrum is used to identify different elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
